The archaea developed rather differently from the Ech driven bacteria. I want to look at them from the energetic point of view so it seems reasonable to start with this image from the first of the Life series posts, back in February:
This has CH3-SH (used at two points in the process) driving acetate formation for cell carbon generation. We know this works because Huber and Wächtershäuser demonstrated the abiotic generation of activated acetate from CO and CH3-SH in the presence of an FeS/NiS slurry. No enzymes, no cofactors, no structure. Energetically, it works. I now want to speculate wildly about other uses of CH3-SH in the development of methanogens and the evolution of methyltranseferase, the archaeal alternative to Ech. Let's get rid of the carbon fixation doodles. The location of CH3-SH might need to change as ideas develop:
In the background to the following speculation we still have a FeNi hydrogenase "visiting" a proton channeling pore to generate reduced ferredoxin using a localised low pH region just inside the cell. In the archaea I'm assuming there is no preformed Na+ pump because the protein translocating precursor never jammed up so never pumped Na+ ions. There was no drop in intracellular Na+ and the FeNi hydrogenase never attached firmly to the proton pore in the membrane. Interestingly the methanogens do still have a cytoplasmic FeNi hydrogenase (or NiFe in this illustration) passing electrons down FeS wires. Instead of dropping them straight on to ferredoxin to generate reduced Fd2- using the proton gradient of the cell wall conducted through a membrane pore (as per proto Ech), pairs of electrons are split at an FAD. Half do the up hill job of generating Fd2- and the other half tumble down hill to the easy target of heterodisulphide. Energy from the latter down hill reaction is used to allow the up hill Fd reduction and gets you out of the need for a membrane proton gradient. I do wonder if this is the same FeNi hydrogenase of proto Ech but here diverted to electron bifurcation. This is from Buckel and Thauer's fantastic paper Energy conservation via electron bifurcating ferredoxin reduction and proton/Na+ translocating ferredoxin oxidation:
That's what happens today. What might have been the core process when metabolism was less refined? Here's a scheme with FeNi hydrogenase (in Hdr, heterodisulphide reductase) using CH3-SH as the electron acceptor for a crude version of electron bifurcation:
Under circumstances of freely available CH3-SH there is no need to conserve sulphur.
The Ni is shown associated with the enzyme which generates all of the biological methane ever produced on earth, methyl coenzyme-M reductase. Nowadays the Ni is bound in the lovely and highly complex coenzyme F430 (an interesting read if you have access):
Here it is in the step producing methane:
Sulphur is no longer a disposable commodity and it is recycled via CoM as a loop in combination with another sulphydryl based coenzyme, CoB.
CoM provides the -CH3 and CoB provides the -H to generate methane. The reaction joins the two coenzymes together to give the mixed, disulphide bridged, heterodisulphide. This is the modern electron acceptor in the Hdr electron bifurcating hydrogenase. It actually accepts a pair of electrons to give CoM-SH and CoB-SH:
The CoM-SH is regenerated to CH3-S-CoM by the methyltransferase shifting a -CH3 from CH3-H4MPT. Ultimately the energetics of the cell is determined by the availability of CH3-SH analogue CH3-S-CoM controlling electron bifurcation at Hdr:
Next let's take out the electron bifurcation system having established a central role for CH3-S-CoM to energetics control and add in the proton port being used to generate Fd2- using the vent H+ gradient:
And add in the antiporter for Na+ ions:
At this point there is some benefit to converting a protein-translocase to a Na+ driven ATP synthase. Consider that it is Na+ ions that are stabilising the membrane section of the translocase so it seems logical to accept a sudden Na+ gradient as the force pushing inwards to reverse the translocase to generate ATP. So we need the A1-A0 ATP synthase adding in to the diagram:
Now a proton gradient is once again driving a Na+ coupled ATP synthase and all is hunky dory for the cell. Excess ATP synthesis can be regulated by CH3-S-CoM inhibiting Na+ antiporting:
When the vent proton gradient fails all that is needed is for CH3-H4MPT to take over from the vent proton gradient. The transfer of the methyl group from H4MPT to CoM is exergonic and is used to drive a conformation change in B12 which ejects the Na+ ion. Lots of detail from Thauer again at The Na‡-translocating methyltransferase complex from methanogenic archaea.
Which simplifies to this:
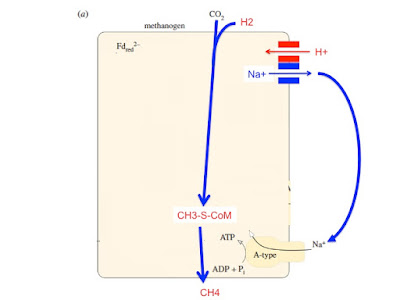
Giving a speculative journey leading to how we might have ended up here:
In modern methanogenic archaea Na+ energetics have been carried forward to today. Protons still look like an add on to me. Of course the question posed from here is how similar is the Na+/H+ antiporter of the methanogens to the NouH and NuoL combination in the bacteria, incorporated in to the base of complex I.
Not surprisingly I haven't found anyone crazy enough to float this idea. This is what Thauer has to say about Na+ translocating pores and aspartic acid within the pore channel:
"The second reason for the proposal is that only MtrE has a transmembrane helix with an aspartate residue (Fig. 1), the sequence of this helix in the MtrE subunit from all methanogens being highly conserved: 168-IWGITIGAIGSSTGDVHYGAER-191. An aspartate residue in a transmembrane helix has been shown to be essential for sodium ion translocation as catalyzed by the L-subunit of oxaloacetate decarboxylase from Klebsiella pneumoniae [62]. An aspartate residue is also conserved in the transmembrane helix of the sodium ion-translocating glutaconyl-CoA decarboxylase from Acidaminococcus fermentans and of the sodium ion-translocating methylmalonyl-CoA decarboxylase from Veillonella parva and Propionigenium modestum [63]".
Here is NuoH from complex I with the aspartic acid at D213 picked out in red:
Which rather implies that NuoH, rather than NuoL, was the Na+ part of the antiporter, assuming the membrane portions of methyltransferase and Ech derivatives are distant relatives of the same ancestral protein...
Peter
Subscribe to:
Post Comments (Atom)

















No comments:
Post a Comment